In the now ubiquitous computer-generated image of the SARS-CoV-2 virus that has ricocheted around the world in the year since it was created at the Centers for Disease Control and Prevention, it is the virus’s “spike” proteins that catch the eye. Rendered in lurid red-orange, the spikes bristle like a dense forest on the barren, gray surface of a distant planet, in both color and form embodying the dread and sorrow that COVID has unleashed upon humanity.
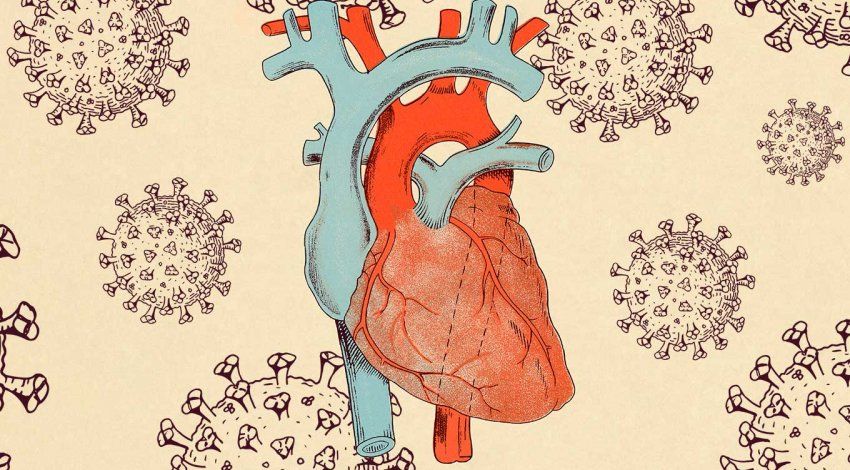
But the spikes of SARS-CoV-2 would be no more than a biological curiosity were it not for a less-familiar protein called ACE2, which provides the portal through which the virus enters our cells to hijack them for its own ends.
For decades before the pandemic, biologists typically thought about ACE2 in terms of its crucial role in regulating blood pressure. It was in this context that the protein was discovered in 2000 by Susan Acton, PhD ’91, who was then leading a team searching for new cardiovascular drugs at Cambridge, Mass.-based Millennium Pharmaceuticals.
Acton, now senior director of research at Artugen Therapeutics, a Concord, Mass., company that develops microbiome-based drugs, says she was somewhat startled when mentions of ACE2 began popping up in the news once COVID took hold.
“I didn’t hear about it in the science literature – it was in the media,” Acton says. “It was surprising, and, I have to admit, a little bit exciting to have been involved early on in something that’s pretty important now. There is enjoyment in saying, ‘Hey, yeah, I discovered that.’”
It was surprising, and I have to admit, a little bit exciting, to have been involved early on in something that’s pretty important now.”
But ACE2 is just one notable protein on Acton’s trophy shelf: In 1996, as a postdoc at the Massachusetts Institute of Technology (MIT), she discovered the HDL (high-density lipoprotein) receptor. It was “a big deal at the time,” she says, “because it’s the major player in removing cholesterol from the body.
“I got a first-author Science paper out of that, which was more than I ever imagined,” Acton recalls. “That finding blew open a whole field of research, and it’s nice when all of sudden a lot of people are working on a protein you discovered. And it’s the same with ACE2 – now a lot of people are working on it.”
Acton has known ever since she was an undergraduate at the University of Michigan that she wanted to work in the pharmaceutical industry, and she says UCSF’s School of Pharmacy provided a perfect springboard to her MIT postdoc and eventual career in drug discovery.
She recalls daily rides on the N Judah light-rail line from her Sunset District apartment to UCSF’s Parnassus Heights campus. There, in the lab of Frances Brodsky, DPhil, now director of the Division of Biosciences at University College London, she and her colleagues worked in close quarters. “I think there were three of us at a single bench, which barely fit in a tiny room. But we concentrated on the research, and we learned so much from one another by working so tightly together.”
When Acton joined Millennium following her postdoc, it was just a bit bigger than a startup – about 100 employees – and gene-sequencing technology was coming into its own.
In a collaboration with scientists from Eli Lilly, Acton’s group at Millennium sequenced a series of genes from the cardiac tissue of a woman with heart disease, to see whether any of her genes might be similar to known disease genes.
A sequence came up that was 42% identical to a previously discovered protein called ACE, which generates peptides – short amino acid sequences – that constrict blood vessels. Acton and her colleagues, including Mary Donoghue, first author on the team’s publication, soon established that the gene they discovered produces a protein that likely counteracts ACE by generating its own peptides that relax blood vessels. Acting together, these complementary proteins keep blood pressure in balance.
Like all explorers, Acton and her team had the happy task of naming the new discovery, and that decision was shaped by pure practicality. “We debated what to call this protein: ‘ACE homolog’ – which we’d abbreviate ‘ACE-H’ – or ‘ACE2.’ I just found ‘ACE-H’ harder to say, so I decided on ‘ACE2,’ because if I were presenting, I’d want it to be easy to say.”
As it happens, a research group at the University of Leeds in England was pursuing the same protein and nipping at the Acton group’s heels, and the two teams published their findings almost simultaneously. “But interestingly,” Acton says, “the Leeds group called it ‘ACE-H,’ and our name stuck.” Millennium, subsequently acquired by pharma giant Takeda, was later granted the patent for the ACE2 sequence.
“We didn’t know when we first sequenced the gene whether ACE2 was a ‘good’ protein or a ‘bad’ protein, but at that point, I had a little bit of a track record of finding good proteins. Like the HDL receptor, it turned out that ACE2 may be more protective than bad for cardiovascular health,” Acton says. “When it comes to the coronavirus, of course, it’s clearly a negative player in that it assists the virus.”
When asked whether she ever feels the urge to tug the sleeve of a stranger to crow about discovering a protein that plays a central role in the global pandemic, Acton says she has discreetly shared her story with family and a few close friends and coworkers, but she hasn’t followed up with the few journalists who’ve made the connection.
“I’m not the kind of person who wants or likes publicity,” Acton says. “But UCSF’s my alma mater, and I loved my time there, so I’ll make this one exception!”
Digital illustration: Stephanie Koch; source images: CDC, Steve Babuljak